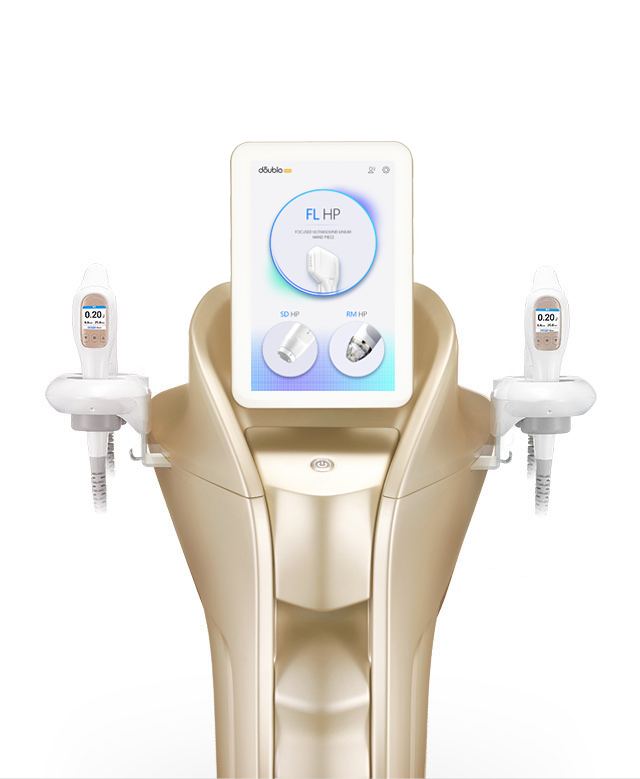
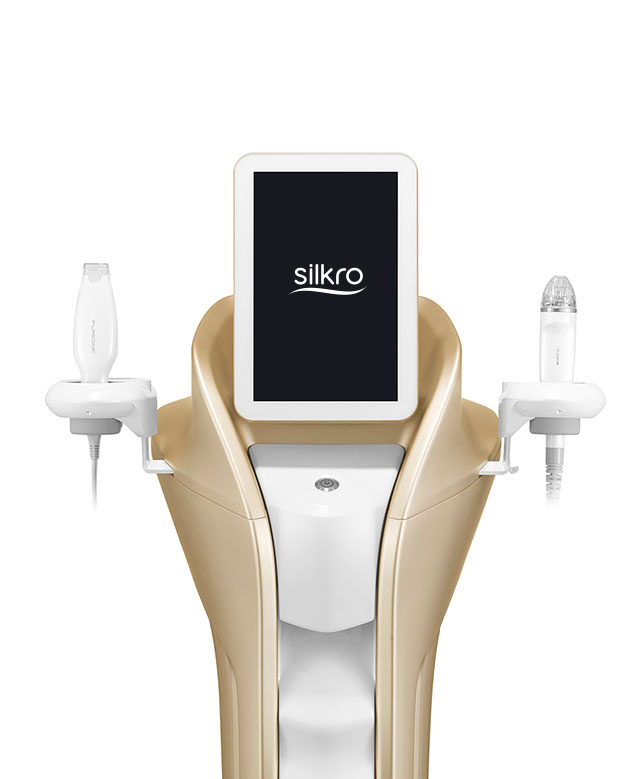
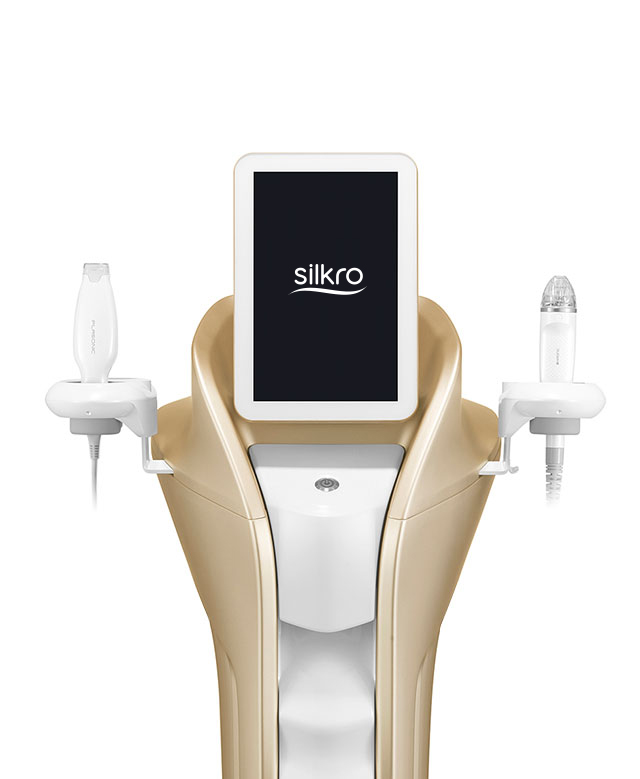
BEST-SELLING PRODUCTS
News MORE
HIRONIC Signs Exclusive Supply Agreement with EMAGE Medical in the U.S. for “Next-Generation RF Combination Platform”
26-05-18
HIRONIC Wins Service Customer Satisfaction Award Chosen by Consumers for the Ninth Consecutive Year
26-05-18
HIRONIC Obtains Official Domestic Medical Device Approval for Its Needle-Free Injector SYNERJET PRO®
26-05-18
HIRONIC Strengthens North American Market Presence Through Participation in ASLMS
26-05-18
HIRONIC GATHERS GLOBAL PARTNERS, STRENGTHENING COLLABORATION AT PARTNERS MEETING
26-04-20